Last year, it was around this time that the everyday cases of COVID-19 started increasing rapidly across India. As practices such as wearing masks and social distancing were still not adopted religiously by the citizens, the fear of community transmission was always lurking. When the complete lockdown came into effect, restrictions were placed on the free movement of people and goods which saw priorities changing for different sections of the society.
While consumers were looking to stock as many supplies as possible, the manufacturers were trying hard to keep up with the panic buying. Meantime, the authorities had their sight set on testing as many symptomatic people and isolate them if found COVID positive. But due to the severity and expansion of COVID -19 across the globe, there was an acute shortage of testing kits and the government had to rely on local manufacturers to shore up the supplies.
The Challenge
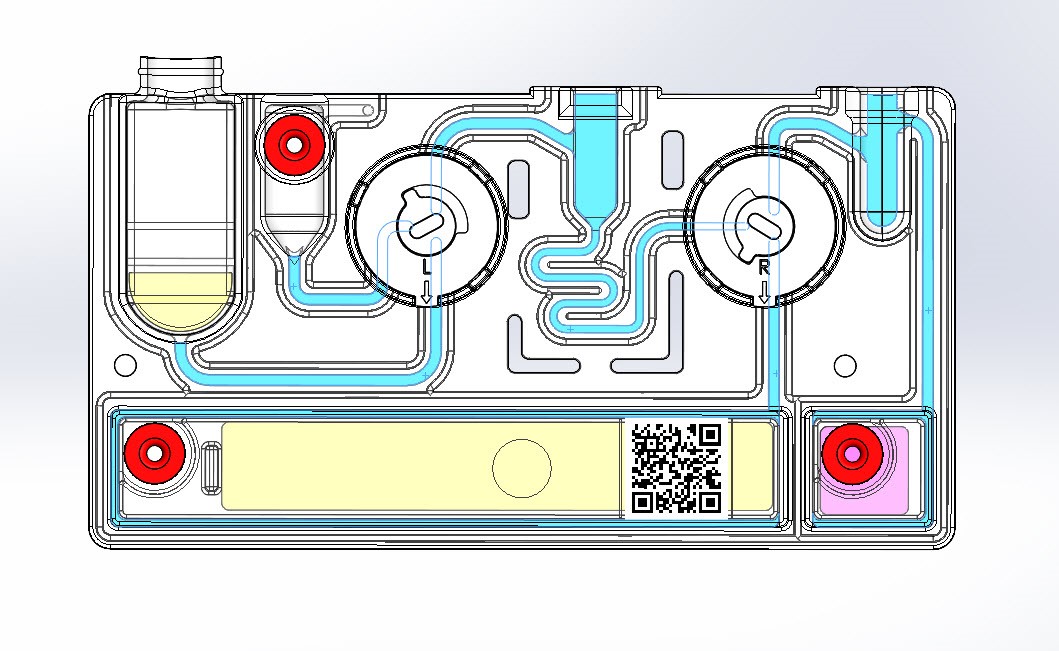
With limited skilled manpower and high demand for effective COVID-19 testing kits, a leading medical diagnostic maker was running against the clock to meet the quality and quantity parameters. The manufacturer was looking to replace the manual quality inspection process of the ‘cartridge’ (a key component used in the COVID-19 testing kit) with an automated one. The key inspection objective was to ensure that the channel of the cartridge is free from any impurity or bur which would obviously hamper the quality and effectiveness of the testing kit.
OMRON pitches in
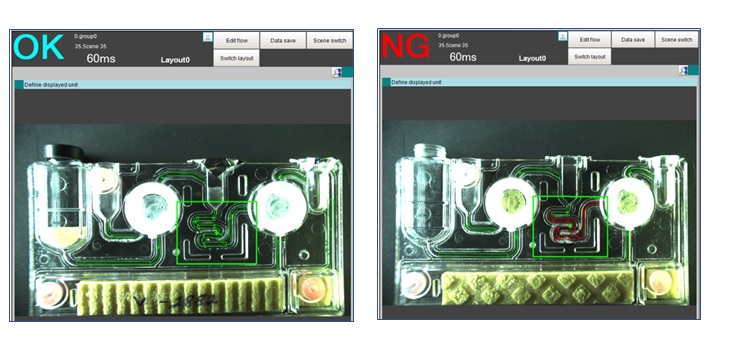
Having already been introduced to OMRON’s innovative automation portfolio, the manufacturer shared the project details with OMRON technical team who aggressively conducted trials at the customer site even during the lockdown. During the trials, it was observed that OMRON’s Vision System was able to identify impurities present in the channel and it worked in tandem with OMRON PLC that would collect data of rejected cartridges. Another benefit was the reduction of inspection time for each cartridge which came down to approx 150 milliseconds as opposed to 500 ms during the manual checking.
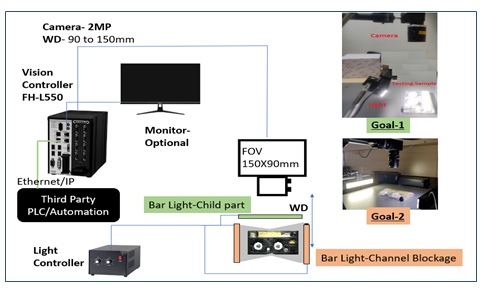
The solution architecture
After concluding the trials and analyzing the results, team OMRON shared its detailed recommendations with the customer which included setting up the correct architecture for the application along with suggestions on a conducive working environment. The customer was really satisfied with the efforts put in by the OMRON team in a very short span of time and happily agreed to bring OMRON onboard as an automation partner.


Way ahead
India is currently witnessing the 2nd wave of COVID-19 infections with over 300 thousand active cases and over fifty thousand new infections each day. The need to have a quicker and accurate diagnosis is greater than ever before as it strengthens primary healthcare and provides patients with early detection and subsequent treatment. Considering India’s huge population, diagnostics makers are under immense pressure to produce high-quality testing equipment in huge numbers in a shorter span of time. This is where choosing the right inspection technology and a reliable automation partner becomes very important and OMRON will continue to contribute to Indian society.
To learn more, visit www.omron-ap