Sensors are utilized in electronics-based medical equipment to change different various forms of stimuli into electrical signals for analysis. Sensors can build the insight of medicinal equipment, for example, life-supporting inserts, and can empower bedside and remote observing of essential signs and other well-being factors. An aging and expanding population is accelerating the development of new and different types of medical equipment, including various sensors used inside both equipment and patients’ bodies. Healthcare organizations want real-time, reliable, and accurate diagnostic results provided by devices that can be monitored remotely, whether the patient is in a hospital, clinic, or at home.
Key Sensors and Applications
Pressure sensors: are used in anesthesia delivery machines, oxygen concentrators, sleep apnea machines, ventilators, kidney dialysis machines, infusion and insulin pumps, blood analyzers, respiratory monitoring and blood pressure monitoring equipment, hospital beds, surgical fluid management systems, and pressure-operated dental instruments.
Temperature sensors: are used in anesthesia delivery machines, sleep apnea machines, ventilators, kidney dialysis machines, blood analyzers, medical incubators, humidified oxygen heater temperature monitoring and control equipment, neonatal intensive care units to monitor patient temperature, digital thermometers, and for organ transplant system temperature monitoring and control. Applications for flow sensors include anesthesia delivery machines, oxygen concentrators, sleep apnea machines, ventilators, respiratory monitoring, gas mixing, and electro-surgery, in which high-frequency electric current is applied to tissue to cut, cause coagulation, dessication, or destroy tissue such as tumors.
Image sensor: applications include radiography, fluoroscopy, cardiology, mammography, dental imaging, endoscopy, external observation, minimally invasive surgery, laboratory equipment, ocular surgery and observation, and artificial retinas.
Accelerometers: are used in heart pacemakers and defibrillators, patient monitoring equipment, blood pressure monitors, and other integrated health monitoring equipment.
Biosensors: Find applications in blood glucose and cholesterol testing, as well as for testing for drug abuse, infectious diseases, and pregnancy. Magneto encephalography (MEG) and magneto cardiography (MCG) systems use superconducting quantum interference devices or SQUIDs. These highly sensitive magnetometers measure extremely weak magnetic fields and are used to analyse neural activity inside the brain.
Encoders: Can be found in X-ray machines, magnetic resonance imaging (MRI) machines, computer-assisted tomography equipment, medical imaging systems, blood analyzers, surgical robotics, laboratory sample-handling equipment, sports and healthcare equipment, and other noncritical medical devices.
Medical Electronics Market
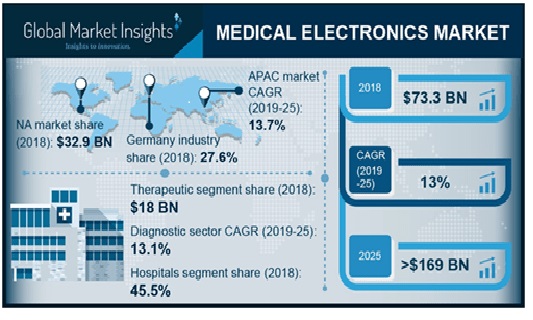
The medical electronics market is estimated to grow from its current market value of more than $73.3 billion to over $169 billion by 2025, according to the latest study by Global Market Insights, Inc.
The study noted rising adoption of technologically advanced medical electronic devices in emerging economies such as India and China will significantly boost the industry growth during the analysis period.
Availability of medical equipment and devices on lease will also escalate its demand among healthcare professionals thereby positively influencing the industry growth, it said.
Rising adoption of technologically advanced medical electronic devices in emerging economies such as India and China will significantly boost the industry growth during the analysis period. Although the medical electronic devices are expensive, several government initiatives and favorable FDI policies will trigger the adoption of these devices among the healthcare professionals. However, stringent regulations enforced on the companies involved in manufacturing of medical electronic devices will restrain the industry growth in the forthcoming years.
Key industry players operational in medical electronics industry include Allengers Medical, Boston Scientific, Carestream Health, FUJIFILM Medical Systems, GE Healthcare, Hitachi High Technologies, Corporation Maxim Integrated, Medtronic, Olympus Corp., Philips Healthcare and others. These players implement several strategic initiatives such as collaborations, new product launch, mergers and acquisitions to acquire potential market share.
Closing Thoughts
Medical devices are taking advantage of the technology convergence. MEMS health sensors made for automobiles are now being used for healthcare devices such as heart monitors and 3D motion tracking. There is a great interest in trying to monitor the activity of people who are at medical risk using remote or implantable sensors, and the insertion and management of artificial devices implanted within the human body is expected to become increasingly common.
This market undergoes constant R&D work. For example, the National Institute of Health (NIH) Roadmap of Nano medicine Initiative expects that, in the next 20 to 30 years, Nano sized implantable solutions will be developed with the ability to receive, store, transmit, and act on information. Continued innovation in sensor components and technology, such as packaging design and device miniaturization, enables a host of new medical applications that save lives and improve quality of life.
Healthcare professionals want real-time, reliable, and accurate diagnostic results provided by devices that can monitor the patient wherever the patient is located. Medical equipment designers want more than just a sensor. They are looking for integrated functionality to provide equipment with improved performance and reliability, and at a lower cost.
Sensors for medical applications are expected to see developments in the following areas: inherent accuracy, intelligence, capability, reliability, small size, power consumption, packaging, cost, and the elimination of lead. Developments are expected to be mainly in the areas of MEMS and nanotechnologies. Market challenges include regulatory compliance, extended product life cycles, reduced product development time, and product safety.