Mercury is harmful even in small amounts. Detecting it currently requires expensive equipment. Researchers are working on a faster and cheaper alternative: a portable sensor that can perform a rapid analysis in the field. The key is finding something small and accurate enough to do the job.
For many years, scientists have studied tiny gold nanorods for making smaller mercury sensors. Recently, a team from the Tyndall National Institute based at University College Cork found that individual gold nanorods could be used to detect mercury with high sensitivity, making them a strong contender for portable analysers. The results were published in the journal Science and Technology of Advanced Materials.
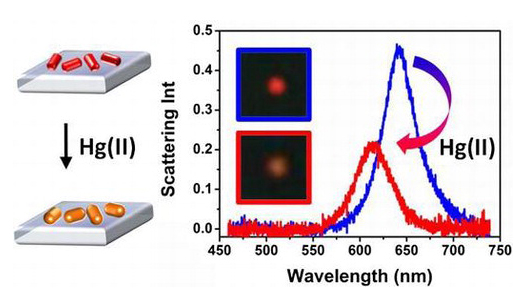
An individual gold nanorod was fixed to a glass slide that was placed under an electron microscope. Using an imaging method called dark-field microscopy, the team studied the composition of the sample by measuring how light scatters off the rod’s surface. A gold nanorod produces a red wavelength pattern, but when it was dipped in a salty solution containing trace levels of mercury, the shape and composition of the rod changed, producing an orange wavelength pattern. The more mercury in the solution, the more the wavelength changed. The nanorods were found to be far more sensitive to mercury than to other metals, including lead, nickel, copper and magnesium.
“The reported linear correlation and high selectivity make this approach potentially suitable for on-site analysis using a miniaturized portable spectrometer,” the study concluded.
However, major hurdles remain. The size and shape of gold nanorods vary from rod to rod, significantly throwing off measurements. Manufacturers must improve their fabrication so they are consistent. Also, pre-purification protocols of the rods will be required before real-world analysis can be reliably performed.